|
Its compounds can be converted to niobium oxide, which is harmless. Niobium doesn't require special disposal. Niobium halides hydrolyze in the presence of moisture and thus will cause burns to the skin. Niobium dust is an eye and skin irritant and a potential fire hazard. Niobium has no known biological role, however elemental niobium on a larger scale is physiologically inert (and thus hypoallergenic) and harmless, being used in jewelry and in some medical implants. The resulting ferroniobium can be further purified. Oxidizers like sodium nitrate can be added to speed up the reaction. Another way involves the aluminothermic reaction a mixture of niobium pentoxide, iron(III) oxide and aluminium:ģ Nb 2O 5 + Fe 2O 3 + 12 Al → 6 Nb + 2 Fe + 6 Al 2O 3 Niobium can be obtained by reducing niobium pentoxide with a reducing agent, such as carbon, hydrogen. Niobium is sold by various chemical suppliers and other metal companies. Niobium has the largest magnetic penetration depth of any element. It is also one of the three elemental Type II superconductors, along with vanadium and technetium. The superconductive properties are strongly dependent on the purity of the niobium metal. Niobium becomes a superconductor at cryogenic temperatures and has the highest critical temperature of the elemental superconductors, 9.2 K. In the presence of air, the surface of the metal develops a protective oxide coating, giving the metal a blueish color. Niobium is a lustrous, grey, ductile, transition metal when pure, but becomes brittle if impurities are present. At higher temperatures it reacts with chlorine and hydrogen, at 200 ☌, while at 400 ☌ niobium reacts with nitrogen. It also reacts with fluorine at room temperature. At room temperature is only attacked by hydrofluoric acid, but hot minerals acids will corrode it. Niobium Atomic Number: 41, Atomic Radius: Atomic Symbol: Nb, Melting Point: Atomic Weight: 92.91, Boiling Point: Electron Configuration: Kr5s14d. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.Niobium is highly resistant to corrosion. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Free Gift for you: Interactive Periodic Table  
If Niobium metal is kept open in the air, it forms the oxide layer of blue, green or yellow color depending on the thickness of the oxide layer.According to USGS, most of the Niobium which is available today, comes from Brazil and Canada.The name Niobium came from the Greek goddess “Niobe” (meaning the daughter of king Tantalus).Around 80% of niobium is used in the production of steel.The former name of niobium (Nb) was columbium (Cb).Interesting facts about niobium element are mentioned below. Hence due to incomplete d-orbitals of Niobium, it forms colored compounds. This emitted radiation has specific wavelengths depending upon the number of electrons present in the d-orbitals as well as elements with which niobium reacts. When this excited electron loses its energy, it again comes back to the lower energy level.ĭuring this phenomenon, it emits an electromagnetic radiation which has the wavelength of visible light. When light is incident on a Niobium metal, its outermost electron gets excited onto a higher energy level. It’s the most plentiful metal found in Earth’s crust. Niobium is a transition metal and it has incompletely filled d-orbitals. Niobium is a chemical element with atomic number 41 in the periodic table. In order to have the complete d-orbitals, there must be 10 electrons in it.īut in the ground state electronic configuration of Niobium, you can see that it has only 4 electrons in d-orbitals.Īnd hence, as Niobium has incomplete d-orbitals, it is considered as a transition metal.Īlso see: Location of transition metals on periodic table (Image) Does Niobium form Coloured Compounds? Protons 41 Neutrons 52 Electrons 41 Symbol Nb Atomic massĢ, 8, 18, 12, 1 Electronic configuration 4d 4 5s 1 Atomic radiusĢ07 picometers (van der Waals radius) 1st Ionization energy 6.759 eV ElectronegativityīCC (Body Centered Cubic) Melting point 2750 K or 2477 ☌ or 4491 ☏ Boiling point 5017 K or 4744 ☌ or 8571 ☏ Density 8.57 g/cm 3 Main isotope 93Nb Who discovered Niobium and when?īecause, there are only four electrons in the d-orbitals. 
Metallic grey State (at STP) Solid Position in Periodic table Niobium Element (Nb) Information Appearance 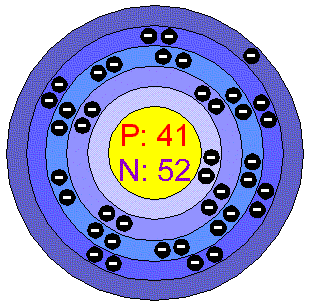
So if you want to know anything about Niobium element, then this guide is for you. The electron configuration of niobium ion (Nb 5+) is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6. In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Niobium element in Periodic table.) During the formation of a bond, niobium donates an electron of the last shell and four electrons of the 4d orbital to form bonds and turns into a niobium ion (Nb 5+ ). This is a SUPER easy guide on Niobium element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
